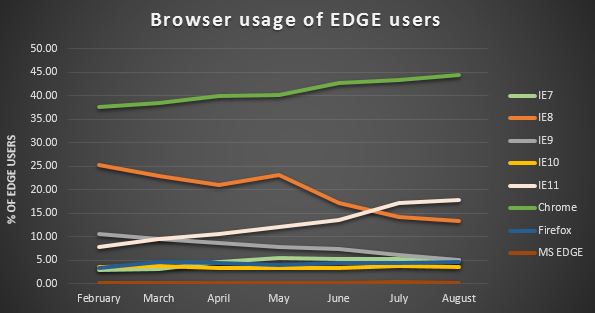
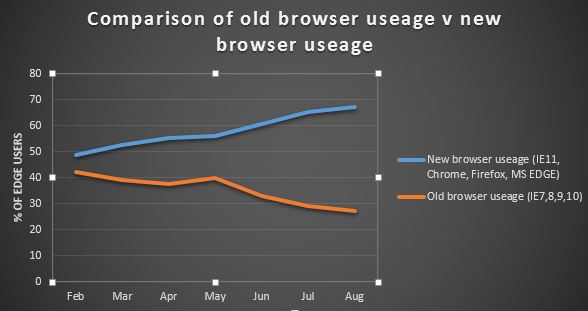
This post has been written by Rebecca Xu, 3CTN- Canada
Successful clinical trials management for the 3CTN Coordinating Centre (CC) and for 3CTN sites requires a flexible system to track trial-related activities. The system has to work well at two levels: for the CC and the sites. The CC needs to have a system to support the identification and communication of portfolio trials to patients and the public. Also, such a system is important for the evaluation of 3CTN against its milestones and deliverables laid out in its business plan. In addition, to demonstrate an increase in recruitment to academic clinical trials in Canada. 3CTN required each participating site to establish an accurate baseline for recruitment. This is used to continuously evaluate the performance and impact of the Network. A flexible system facilitates this evaluation. Among all Canadian cancer centres responding to the initial 3CTN expression of interest, there was a demand for a clinical trial management system (CTMS) to manage the clinical trial projects, track clinical trial activities, staffing, milestones, metrics and finances centrally and in an organized fashion. The optimal solution to track the metrics and research activities is a common platform for all member sites and the 3CTN Coordinating Centre.
3CTN went looking for a CTMS solution. After consultation with a wide variety and number of stakeholders, and following the recommendation of a working group, based on the positive impact in the UK and first Canadian site (i.e. Juravinski Cancer Centre), the EDGE Clinical Research Management System (EDGE) was selected by 3CTN as the preferred system to roll out at the CC and Canadian cancer centres that expressed interest.
The EDGE system, which was developed in the U.K by the University of Southampton and University Hospitals Southampton National Health Service Foundation Trust, was first introduced in 2001. Since then, EDGE has been used by all the U.K. cancer research networks and several other networks established by the Department of Health in the U.K. EDGE provides an innovative cloud-based clinical management system that empowers administrators and investigators to make the most of their research data. EDGE is specifically designed to give a real-time view of clinical research activity within a single hospital or across various research networks.
The CC formally adopted EDGE in September 2014, with the first goal being the tracking of the portfolio. The CC EDGE hosts a comprehensive listing of all 3CTN-endorsed cancer clinical trials. It allows management of the portfolio, including trial application, and timeline tracking of the review and approval process in a clear, efficient and measurable system. In addition to portfolio tracking, the CC can process all data in EDGE and extract the aggregate and site efficiency metrics with ease. It provides a real time and accurate picture of the high impact academic clinical trials across Canada. The national benchmarking data has also proven to be useful to facilitate ongoing process improvement.
EDGE provides an efficient way to collect and process the data without imposing an excessive burden to 3CTN sites. This common platform limits duplication of data entry and ensures ease of data transfer between 3CTN member sites and the CC. For sites that had already adopted a different CTMS prior to 3CTN, EDGE provides portal capability and flexibility to take-in data from the various systems used by sites.
Since the adoption of EDGE by 3CTN, the CC has developed comprehensive implementation supports. These supports include: live demonstrations of the system, project planning, training and first line support to facilitate the roll-out to interested cancer centres. Regional super users were hired to assist with implementation and to ensure sites use EDGE effectively. These super users are situated throughout the Network, and collaborate with the CC to promote best practices, and to determine how EDGE can best be used to benefit all sites across the country.
At the site level, most sites using EDGE are tracking recruitment. Many sites are starting to use EDGE to track regulatory processes and finances for their active recruiting trials, as well as legacy data. The EDGE users are especially pleased with the powerful reporting capabilities of EDGE, which allows them to follow trends in research activities over time. Prior to the implementation of EDGE, sites had numerous files, over a number of file directories to keep track of the progress of trial activation, ethics submissions, contract processes, budgets, and trial metrics (screening and accrual reports). From a logistics standpoint, having one place to access all clinical trial information is very efficient, and ultimately improves patient care as it enables everyone to use the most up to date documents and information. All members of the patient care team are able to see the progress of a trial from delivery of the protocol to trial activation, and the screening and accrual of patients. “EDGE provides us with a wealth of knowledge to know which trials are accruing and which are not and why not” states Richard McClelland from London Regional Cancer Centre. Being a network-customized and web-based application, EDGE has led to better communication and collaboration within the cancer clinical trial community, and across the country.
As a result of CC and site level implementation, the EDGE system is helping to improve the conduct of cancer clinical trials across Canada. It enables reliable and real-time measurement of trial metrics and improves the ability of sites and the CC to analyze and use trial data to advantage which should lead to more efficient activity across the Network.
Source of article from: Canadian Cancer Clinical Trial Network 3CTN Pulse Newsletter February 2017